 
The atomic sizes of non-metals determine the number of other properties.Įlectrical conductivities of non-metals are extremely low. Non-metal atoms are often smaller than metal atoms. Non-metals have the following properties in general. Liquid non metal at room temperature is bromine. Carbon, nitrogen, fluorine, sulphur, and iodine are non-metal dopants. The anionic dopants have a substantial influence on the VB among the non-metals. Non-metals frequently gain electrons when reacting with other compounds, forming covalent bonds, because of their features. High ionization energies and electronegativity are two characteristics of non-metals. In this article, we will learn about non-metals, properties of nonmetals, periodic table metals and nonmetals. 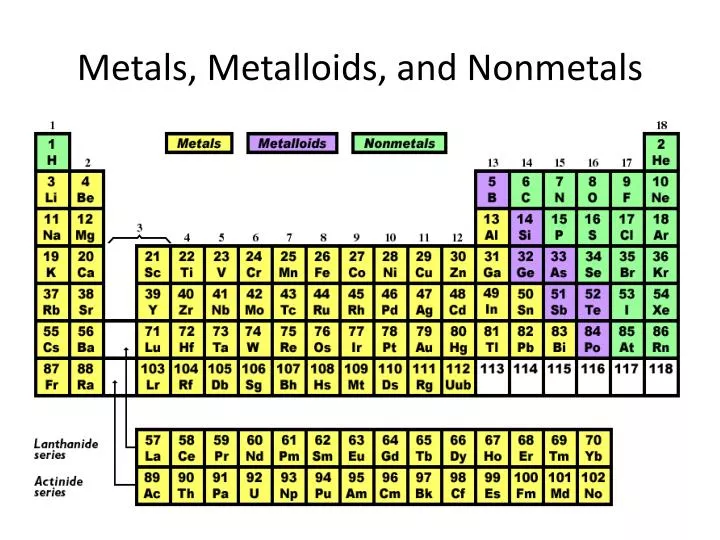
Carbon, sulphur, and phosphorus, for example, are solid at normal temperatures. They are usually gasses, although they can also be liquids. They are excellent heat and electricity insulators. Non-metals are materials that lack all of the characteristics of metals. They often have 4, 5, 6, or 7 electrons in their outermost shell. Non-metals are elements that accept or gain electrons to generate negative ions. It is also available in PDF form to download. They can use it for both revisions and for understanding the topic. Thus, it will be very beneficial for students in their exam preparation. The explanation provided here has been written keeping this in mind. They have knowledge about the kind of challenges students face while learning the Non-Metals topic. The Non-Metals article has been prepared by the expert faculty who have a lot of experience in teaching the Non-Metals topic to students. Thus, students can get most of their doubts cleared on the topic with the help of the Non-Metals article. The most commonly asked questions have also been answered in the FAQs. To explain the topic well several examples have been provided. Thus, it is very important for students to understand the Non-Metals topic really well. The Non-Metals topic is very important for both boards exam and competitive exams. Standard conditions: are standard sets of conditions for experimental measurements defined as a temperature of 273.Students can find the Non-Metals topic explained on this website. Ionic radii: the average distance from the center of the nucleus to the edge of its electron cloud Oxidation state: a number assigned to an element in chemical combination which represents the number of electrons lostĪtomic radius: the average distance between a nucleus and its outermost electron Ionization energy: the energy required to remove an electron from the valence shell of a gaseous atomĮlectron affinity: the energy change that occurs when an atom gains an electronĮlectronegativity: a measure of the ability of an atom to attract the electrons in a bond Valence electron: an outer shell electron that can participate in chemical bonds Malleability: the ability of a metal to be hammered into shapesĭuctility: the ability to be pulled or drawn into wires Non-metals have small atomic radii and large ionic radii.Non-metals have high ionization energy, high electron affinity, and high electronegativity.Non-metals are poor conductors of heat and electricity because they do not easily give up electrons.Metals are good conductors of heat and electricity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
